|
10/29/2022 0 Comments Halogen periodic table 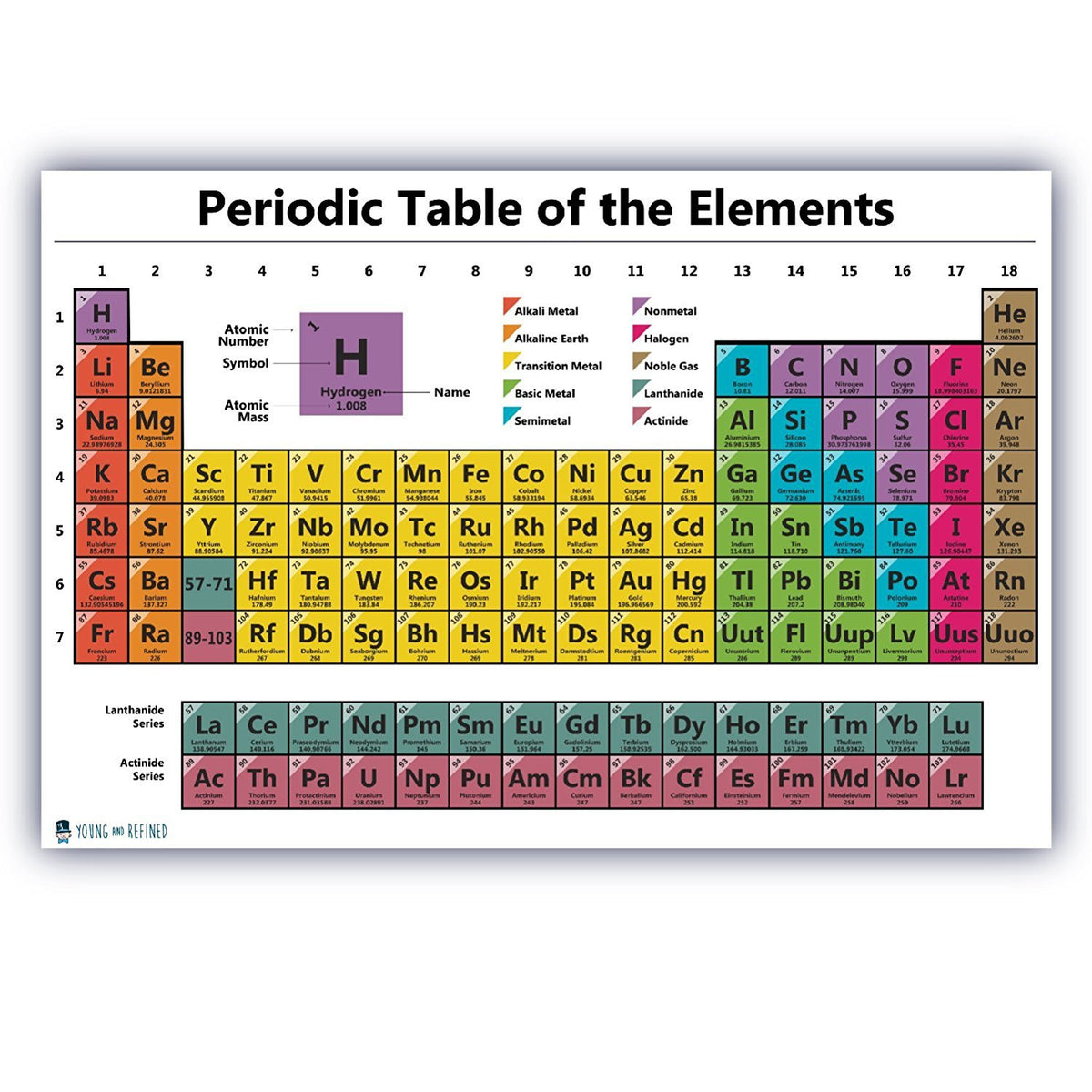
When you look at individual elements on the periodic table, there are several pieces of information that you can find on each square. One group of elements that have nearly indistinguishable chemical properties, called the rare earth elements, is separated out into its own separate space at the bottom of the periodic table. This is because the elements don’t always fit neatly into groups that share the same physical and chemical characteristics. Each of these 7 rows is called a “period.” Finally, you might notice that there are gaps in the periodic table, particularly in the top 3 rows. The orbitals are the regions around the atom’s nucleus where electrons are most likely to appear. When you read the period table in rows going across from left to right, you will find that the elements in each row share the same number of atomic orbitals. 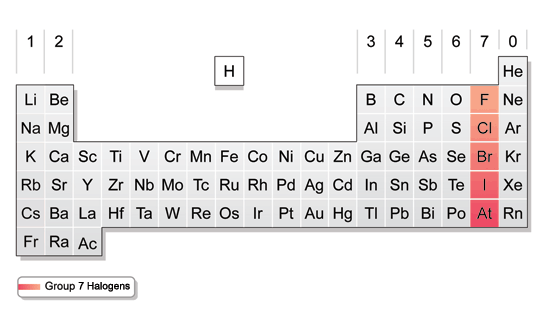
Additionally, related elements are usually color-coded to indicate whether they are metals, semi-metals, or non-metals. Each column in the periodic table, going from top to bottom, represents one of these groups. The elements are also organized into groups, or families, that share similar physical and chemical properties. At the other extreme, an atom of the synthetic element oganesson has 118 protons and an atomic number of 118. For instance, the first element in the periodic table, hydrogen, has an atomic number of 1, because a hydrogen atom has only 1 proton. Each element has its own atomic number, which represents the number of protons in one atom of the element. As you move across the table, the number of protons and the atomic mass of each element increases. 
When you’re reading the periodic table, move across the table from top left to bottom right. But it’s not too difficult to understand once you learn the basics of how it works. The periodic table can seem a little daunting at first. Elements are labeled as semi-metals if they have a mixture of properties of both metals and non-metals.These elements are usually gases at room temperature but may also become a solid or liquid at certain temperatures. Elements are considered a non-metal if they lack luster, don’t conduct heat or electricity, and are non-malleable.Elements are labeled as a metal if they have luster, are solid at room temperature, conduct heat and electricity, and are malleable and ductile.Keep in mind that hydrogen can be grouped with either the Halogens or the Alkali Metals because of its properties, so it may appear on either side of the table or may be colored differently.You’ll notice that metals occur to the left of the table, while non-metals fall on the right. Fortunately, most periodic tables use color to indicate whether the element is a metal, semi-metal, or non-metal. You can better understand the properties of an element by recognizing what type of element it is. One topic of interest for us is how halogens affect the way the molecules that contain them behave, especially a phenomenon called halogen bonding," explained Donald.Distinguish between metals, semi-metals, and non-metals. "They are chemically interesting and versatile: fluorine, for instance, tends to be very reactive yet combining fluorine with carbon in a special way allows Teflon pots to be ‘non-stick’. One of Donald's favorite groups of elements are The Halogens, which include fluorine, chlorine, bromine, and iodine. "The outcomes of that bonding is what we call in chemistry a sandwich compound," said Donald. One paper he's written discusses how the elements beryllium, magnesium, calcium, strontium, barium, zinc, cadmium, mercury, and other elements form bonds to a ring structure made entirely of carbon and hydrogen. "Everything that we can actually see and touch, all summed up with the word ‘matter’, on the earth is some combination of atoms of naturally occurring elements," said Donald.ĭonald's research group investigates molecules involving elements from across The Periodic Table. He can speak in particular to a group of elements known as The Halogens, which he uses in his research as a theoretical and computational chemist. Kelling Donald, Associate Professor of Chemistry and Associate Dean of the School of Arts & Sciences, works with elements across the Periodic Table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
